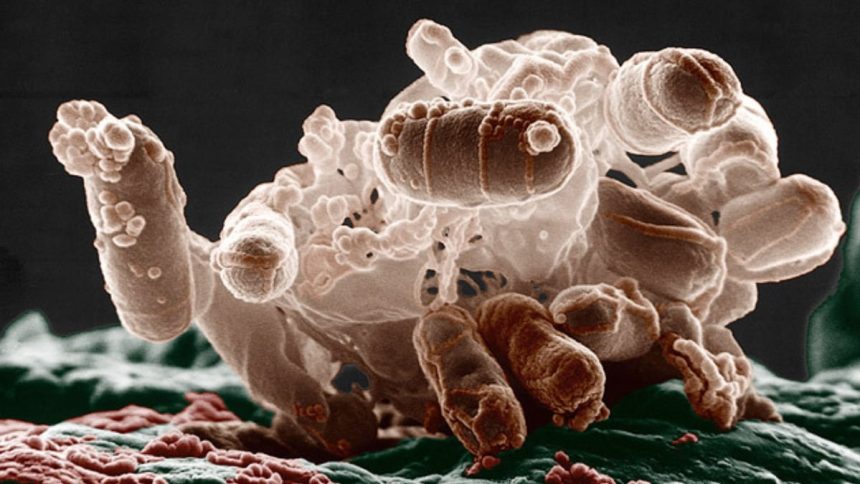
Antibiotics are intended to eliminate harmful bacteria and assist the body in recovering from infections. However, certain antibiotics can also stimulate bacteria to release minute particles that worsen inflammation.
Inflammation is a natural defense mechanism of the body against infections, but excessive inflammation can harm healthy tissues and hinder the healing process. In extreme situations, too much inflammation can become life-threatening.
The particles released by bacteria are known as bacterial extracellular vesicles (BEVs). These microscopic, bubble-like structures carry proteins, toxins, and various molecular signals that affect how the host’s immune system responds.
Bacteria naturally discharge BEVs into their environment to communicate, dispose of damaged cellular material, and interact with host cells.
Despite their tiny size, BEVs can have a significant impact on the human body. When they enter the bloodstream, they can engage with cells lining blood vessels and initiate an immune response.

In certain instances, these interactions can exacerbate inflammation and result in sepsis, a severe condition where the body’s infection response becomes dangerously unregulated, potentially damaging tissues and leading to organ failure.
I am a biomedical engineer investigating the role of bacterial extracellular vesicles in inflammation during infections.
My recent research reveals that some antibiotics prompt bacteria to release significantly more vesicles than others. This discovery suggests that the method by which an antibiotic kills bacteria could also influence the amount of inflammatory material released into the body.
When antibiotics stress bacteria
Antibiotics operate in various ways. Some target the bacterial cell wall, weakening it until the cell disintegrates and dies. Others disrupt essential cellular functions such as protein synthesis or DNA replication, preventing bacterial growth.
Regardless of their approach, antibiotics control infections by killing the bacteria responsible for them.
However, antibiotics also place bacteria under stress, and this stress can lead them to release more extracellular vesicles containing inflammatory molecules.
To study this, I exposed E. coli to several commonly used antibiotics and quantified the vesicles they produced.
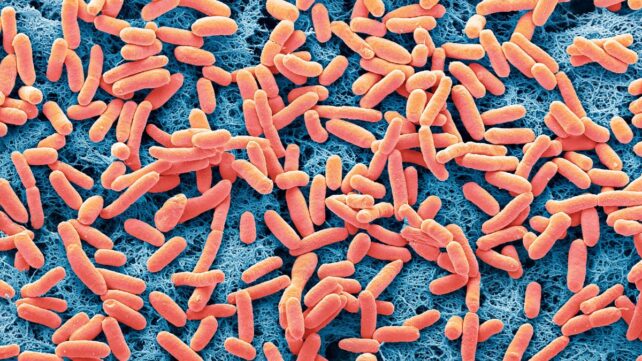
The aim was straightforward: to evaluate how different antibiotics affect vesicle release and determine if the way an antibiotic kills bacteria impacts vesicle production.
The findings revealed that antibiotics do not equally affect the vesicles produced by bacteria.
Antibiotics targeting the bacterial cell wall, such as the widely used beta-lactams, resulted in a notable increase in vesicle production. Conversely, antibiotics acting on protein or DNA processes had a much smaller impact.
This difference probably reflects how bacteria respond to damage. When their cell wall is compromised, bacteria might release more vesicles to shed damaged material or adapt to stress. These vesicles’ inflammatory molecules can further stimulate the immune response.
This raises a critical question: Could some antibiotics inadvertently amplify inflammation and worsen an infection?

While my research does not prove that antibiotics directly cause infections, it suggests that the type of antibiotic could influence not only how effectively bacteria are eradicated but also how the body responds to the infection.
Further research is necessary to comprehend how these bacterial responses affect patients during severe infections, such as sepsis.
Why this matters for treating infections
It is crucial to recognize that antibiotics remain among the most effective and lifesaving interventions in modern medicine. This research does not imply they should be avoided.
Instead, it highlights that bacteria are not passive targets. They actively respond to treatment, and those responses can have additional effects on the body.
Understanding how bacteria react to antibiotics could aid researchers and clinicians in evaluating how different treatments impact both infection and inflammation. In scenarios where controlling inflammation is vital, such as severe infections, these differences may be particularly significant.
Related: Gut Inflammation Link to Alzheimer’s Disease Confirmed Once Again
This research also signifies a wider shift in how scientists view infection. Instead of solely focusing on eliminating bacteria, researchers are increasingly examining how bacteria communicate, respond to stress, and interact with the human body.
As scientists continue to investigate bacterial behavior under antibiotic pressure, it becomes evident that treating infection is not only about stopping bacterial growth but also about understanding the signals bacteria leave behind.![]()
Panteha Torabian, Ph.D. Candidate in Biomedical and Chemical Engineering, Rochester Institute of Technology
This article is republished from The Conversation under a Creative Commons license. Read the original article.