Understanding the New Molecular Clock for Predicting Alzheimer’s Onset
A groundbreaking new ‘molecular clock’ has been developed by researchers to predict the onset of Alzheimer’s symptoms years in advance, utilizing established blood markers. This innovation could significantly improve support for those affected by the disease and revolutionize the way we approach Alzheimer’s research and treatment.
Alzheimer’s disease is a complex neurodegenerative condition with a timeline that can span 10–30 years from the first biological signs to the manifestation of cognitive symptoms. Scientists have long been puzzled by the variability in this timeline and are eager to uncover the factors that influence disease progression.
The newly developed molecular clock aims to narrow down the window during which individuals might start experiencing memory problems to a three or four-year period. While there have been previous predictive tools for Alzheimer’s onset, this molecular clock stands out for its simplicity and efficiency.
The team behind this innovative tool, led by researchers at Washington University in St. Louis (WashU), envisions its utility in group-level studies initially, with the eventual goal of personalized prediction for individual patients.
Neurologist Suzanne Schindler from WashU highlights the significance of using blood tests for predicting Alzheimer’s symptoms, citing their cost-effectiveness and accessibility compared to traditional imaging and fluid tests.
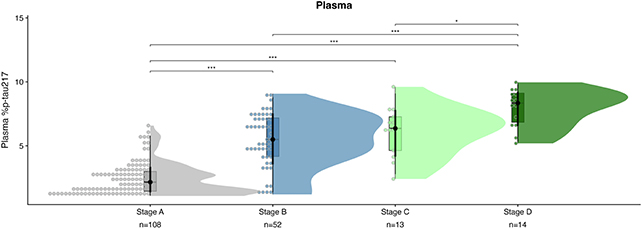
The molecular clock relies on measuring levels of the p-tau217 protein in the blood, a biomarker already used for Alzheimer’s diagnosis. By leveraging mathematical modeling, researchers have unlocked its potential to forecast symptom onset.
Studies have shown that p-tau217 levels in the blood mirror the accumulation of tau and amyloid-beta proteins in the brain, key indicators of Alzheimer’s pathology. Analyzing data from 603 individuals over several years, the researchers established formulas linking p-tau217 levels to cognitive decline onset.
Notably, the research suggests that younger individuals exhibit a longer interval between biomarker elevation and symptom onset, implying a protective mechanism in younger brains against neurodegeneration.
Lead author Kellen Petersen likens the accumulation of amyloid and tau proteins to tree rings, emphasizing the predictive power of these biomarkers in determining Alzheimer’s symptom onset.
While the current accuracy of the molecular clock is best suited for group analyses, its potential for individualized care and clinical trial optimization is promising. By identifying individuals at higher risk of symptom development, researchers can streamline trial design and monitor disease progression more effectively.
The research, published in Nature Medicine, marks a significant leap forward in Alzheimer’s prediction and underscores the importance of early intervention and personalized treatment strategies in combating this devastating disease.





