The potential advantages of GLP-1 drugs are expanding, as a new review primarily of preclinical studies connects these popular diabetes and weight-loss medications to reducing the molecular signs of dementia.
Researchers from Anglia Ruskin University in the UK reviewed 30 published studies conducted on cell cultures and lab animals.
They specifically examined the effects of four different GLP-1 drugs on the harmful accumulation of amyloid-beta and tau proteins in the brain, which are markers of Alzheimer’s disease.
Among the preclinical studies analyzed, 22 indicated a reduction in amyloid-beta plaques, and 19 showed a decrease in tau tangles. These abnormal protein masses are believed to harm and kill neurons in Alzheimer’s disease, though other factors are also being considered.
We are far from confirming that GLP-1 drugs like Ozempic and Wegovy can reduce the risk of dementia, especially since the review only identified two small human trials.
Nevertheless, increasing evidence from cell and animal models of the disease suggests a potential connection.
“This new review provides one of the most comprehensive analyses so far of how GLP-1 drugs interact with the underlying mechanisms of Alzheimer’s,” says physiologist Simon Cork.
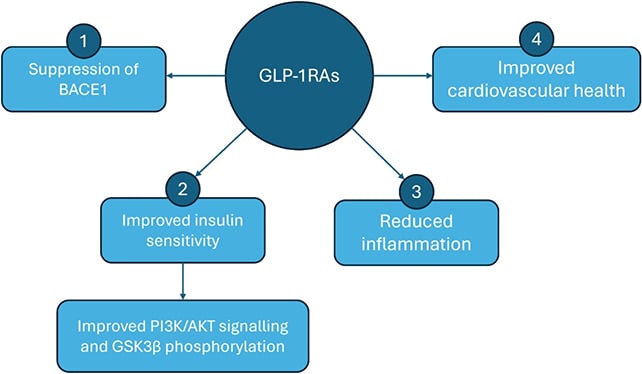
“Our study highlights several biological pathways by which GLP-1 drugs may influence Alzheimer’s, including reducing inflammation, improving insulin signaling in the brain, and altering enzymes involved in the production of amyloid-beta.”
GLP-1 drugs derive their name from the glucagon-like peptide-1 (GLP-1) hormone they imitate. They are technically GLP-1 receptor agonists because they act on the same cell receptors as GLP-1, triggering similar responses—slowing digestion, stimulating insulin release, and suppressing appetite.
These drugs are widely recognized by brand names such as Wegovy, Ozempic, and Mounjaro. However, the key elements are the active ingredients: semaglutide, liraglutide, exenatide, and dulaglutide, which were the focus of this review.
Liraglutide was the most extensively represented active ingredient in the review and proved the most consistent in reducing both amyloid-beta and tau to safe levels.
Exenatide had the smallest impact in the data analyzed, though it still showed a connection with reduced amyloid-beta and tau in some studies.
The review also examined two small clinical trials, which produced mixed results.
In one trial, brain cell metabolism was preserved in one trial, and the other observed reduced amyloid-beta in extracellular vesicles. However, neither trial found that GLP-1 drugs reduced the build-up of amyloid-beta in the brain or prevented cognitive decline.

“Whilst human studies demonstrating an impact on cognitive decline are still lacking, the current evidence points towards these drugs having a preventative effect, rather than in patients with established cognitive impairment,” says Cork.
Previous research has indicated that individuals on GLP-1 medications are sometimes less likely to develop dementia.
However, other studies involving people with early Alzheimer’s or mild cognitive impairment have shown negative results: One study published last year found no association between semaglutide and a slowing of cognitive decline.
We also know that both obesity and diabetes, the primary conditions targeted by GLP-1 drugs, have connections to dementia. Deciphering the various mechanisms at play and their effects on dementia risk will require more time.
Further investigation is needed to understand how GLP-1 medications might protect against toxic protein build-up and dementia. Researchers propose that reductions in inflammation and protein production, along with improved insulin signaling, might play a role.
Related: Scientists Reveal The Optimal Amount of Sleep to Lower Dementia Risk
“With more than three-quarters of preclinical studies showing reductions in amyloid-beta or tau, and early indications from human studies, GLP-1 drugs continue to be promising candidates for future Alzheimer’s prevention trials,” says Cork.
“Larger, early-stage clinical trials are now needed to determine whether these promising signs actually translate into tangible benefits for patients.”
The research has been published in Molecular and Cellular Neuroscience.