Atrial fibrillation is a heart rhythm disorder affecting millions, causing the heart’s upper chambers to beat irregularly rather than in a coordinated pattern. While symptoms like palpitations, fatigue, or breathlessness can be mild for many, the most significant risk is experiencing a stroke.
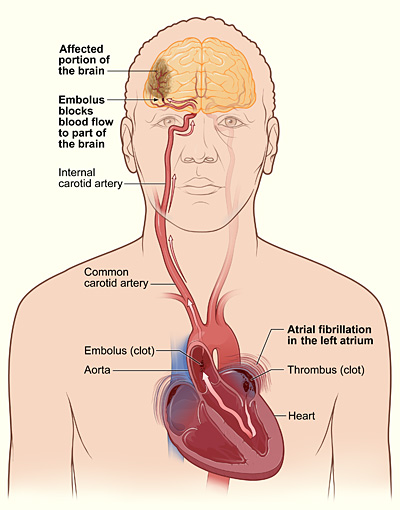
The heart contains a small pouch known as the left atrial appendage. When the heart rhythm is irregular, blood can accumulate in this pouch instead of flowing smoothly, leading to clot formation. If a clot dislodges and travels to the brain, it can obstruct blood flow and result in a stroke.
Having atrial fibrillation increases the likelihood of a stroke by about five times. Researchers have been exploring ways to mitigate this risk by addressing the issue of the left atrial appendage.
Researchers have recently introduced a potential solution—a new technique, currently tested only in animals, involving the injection of a magnetically guided liquid into the heart, which hardens to seal the pouch permanently. Initial tests on rats and pigs indicate this method could potentially reduce stroke risk in people with atrial fibrillation.
Current treatments for atrial fibrillation, such as anticoagulants, are effective but not without drawbacks. These blood-thinning drugs lower stroke risk by reducing blood’s ability to clot. However, they raise bleeding risk, posing danger, especially to older adults or those with conditions like stomach ulcers, hypertension, liver or kidney disease, and cancer.
Some patients cannot tolerate anticoagulants or have to discontinue their use due to bleeding complications. An alternative treatment is the left atrial appendage occlusion, where a small device is implanted to block the appendage. Delivered via a catheter, these devices expand like a tiny metal umbrella to seal the opening.
While these devices can work well, they are not flawless. The appendage’s shape and size vary significantly between patients, making it challenging for rigid implants to create a perfect seal. Blood can sometimes leak around the edges, leading to clot formation on the device’s surface. The device’s anchoring parts can also harm heart tissue.
The newly proposed technique offers a different approach. Instead of using a rigid implant, researchers inject a magnetically responsive liquid, or magnetofluid, into the left atrial appendage via a catheter. An external magnetic field guides and holds the fluid in place, filling the entire appendage despite the blood’s circulating force.
In minutes, the liquid interacts with water in the blood, forming a soft “magnetogel” that seals the cavity. Starting as a liquid, the material adapts precisely to each patient’s left atrial appendage’s irregular shape, potentially creating a more complete seal than traditional rigid devices.
The gel also seems capable of integrating with the heart’s inner lining, forming a smooth surface that might reduce clot formation.
Encouraging early results
The technique has only undergone animal testing so far. Researchers initially tested it in rats and then advanced to experiments in pigs, marking a significant step in cardiovascular research.
In the pig study, the magnetogel remained stable inside the appendage for 10 months without clot or leakage evidence. The heart’s inner lining grew over the gel’s surface, forming a continuous, seemingly healthy layer.
Compared to conventional metal occlusion devices in pigs, the magnetogel produced a smoother lining and avoided tissue damage linked to anchoring barbs. Importantly, researchers did not observe harmful biological effects in the animals.
Pigs are commonly used in cardiovascular research because their hearts closely resemble human hearts in size, structure, and function. Demonstrating the magnetofluid’s safe functioning in pig hearts provides valuable proof-of-concept but does not yet assure the technology’s safety or effectiveness in humans.

Despite promising outcomes, the technique is still in the experimental phase. Researchers need to prove long-term safety, refine the material delivery, and ensure it behaves predictably in larger animal studies before human trials can begin.
Challenges remain, such as the magnetic material’s impact on MRI heart scans, which can obscure parts of the heart. These issues must be resolved before the technology can be implemented in patients. Additionally, medical devices undergo extensive testing, so it may take years before this method becomes a viable treatment option.
If proven safe and effective in humans, the technology could offer a new way to protect individuals with atrial fibrillation from stroke. A catheter-delivered liquid seal might serve as an alternative for patients who cannot tolerate anticoagulant drugs and could address some limitations of current occlusion devices.
Related: Athletes Have a Mysteriously Higher Risk of Irregular Heartbeat
With atrial fibrillation affecting tens of millions of people worldwide, even small advancements in stroke prevention could significantly impact global health.
Currently, the magnetic gel remains a laboratory innovation and not a clinical treatment, but it showcases how materials science and biomedical engineering are paving new ways to tackle one of cardiology’s enduring challenges.
David C. Gaze, Senior Lecturer in Chemical Pathology, University of Westminster
This article is republished from The Conversation under a Creative Commons license. Read the original article.